PRF IN SPORTS MEDICINE & RHEUMATOLOGY
Sports-related issues:
Pain in joints, tendons, ligaments, and muscles, often arise from inflammation affecting the somatosensory nervous system 1. An injury, long term overload, or arthrosis caused by long term wear on the joint, can result in a chronic inflammatory state, in which recruited macrophages stabilizes a continuous pain-inflicting stimulation of neurons 2. This inflammatory state is initiated by the release of soluble pro-inflammatory cytokines and chemokines which further activate surrounding cells 3–7 and recruit circulating leukocytes, including macrophages, to the site of injury 8–10 or arthrosis.
The macrophages are, in this sense, considered to be a regulator of inflammation and neuropathic pain 5,10,11. Through the synthesis and release of inflammatory mediators and interactions with neurotransmitters and their receptors, the immune cells form an integrated network with the somatosensory system, coordinating the immune response, tissue healing and modulates the sensory pathways of pain 8.
Traditional therapies:
Traditional therapies in sports medicine and rheumatology are typically NSAIDs, mid- or short-acting steroids, hyaluronic acid, or other GAGs, in combination or as single drugs. These drugs are merely managing the symptoms by reducing inflammation and pain, and do not aim to cure or reverse the injuries caused by the ongoing degenerative disease or chronic state.
The capacity to modulate inflammation, influence macrophage polarization, and stimulate an acute response leading to accelerated healing and regeneration, puts PRF in a unique position as an autologous biological agent for the treatment of inflammation, pain, and damages in joints and tendons, arising from injuries, arthrosis or arthritis.
.
Platelet Rich Fibrin:
Within sports medicine and rheumatology, the use of PRF can be applied in both conservative treatments as well as arthroscopic and open surgery. PRP has for many years been utilized for knee injections in a wide range of patients including professional athletes 12–17. The more recent introduction (2015) of an additive-free liquid PRF that does not utilize anti-coagulants, and thus will form a fibrin clot upon injection, has been a proposed method to further regenerate cartilage, particularly in the knee 18,19.

WHY PRF IS THE NATURAL CHOICE
IN HUMAN SPORTS MEDICINE
Next Generation
Autologous Blood Concentrate
_____________________
Simplicity and ease of use
One huge advantage of PRF is the easy and quick processing of the final product, making it broadly available to even smaller clinics. The treatments are highly economical based on the simple setup of a centrifuge and proprietary but fairly standardized vacuum tubes.
Safe to use
It is also safe since it´s autologous and without additives. The content of leucocytes furthermore adds to the product’s safety, which is of the most significant importance since iatrogenic infections after joint injections, at best, can be problematic to deal with and risk further degeneration, but in unfortunate situations, such as sepsis, even worse implications are at risk to the health of the patient.
The complete team of regenerative cells
In the PRF, the entire group of white blood cells is included. A large part of the growth factors and the majority of the long-term effects of treatment lies precisely in the inclusion and interactions of the white blood cells 20.

The white blood cells further contribute to a shortening of the inflammatory phase, jumpstarting the proliferative and, thus, the regenerative phase at an earlier time. It is also in this proliferative phase, the formation of new blood vessels (angiogenesis) contributes to increased and better vascularization of the area.
In PRF, the white blood cells, platelets, and various cytokines are all included in a carefully selected fraction, both retained in and stimulated by the fibrin, thereby allowing for the full regenerative potential to be utilized.
Anti-inflammatory Properties
Promoting M2-like Macrophage Polarizing
_____________________
 Modulate Macrophage phenotype
Modulate Macrophage phenotype
PRF is able to modulate the polarization of tissue-residing macrophages30–35, and initiate a shift from the destructive M1-like phenotype towards the regenerative M2-like phenotype29,36.
Intrinsic growth factors
The activated platelets, leucocytes and fibrin matrix in PRF, release an elevated amount of growth factors and cytokines, simulating a larger trauma at the site of injection or on lay7,20–25. In particular, the growth factors PDGF, VEGF, TGF-β and IGF-1 are important in regeneration and wound healing26,27.
Modulation of inflammation
The release of the growth factors native to the platelets and leucocytes of PRF, play a direct role in the modulation of both acute and chronical inflammation, especially by the modulation of tissue-residing activated macrophage polarization 28,29.
Stimulate an acute response
The capacity to modulate inflammation, influence macrophage polarization, and stimulate an acute response leading to accelerated healing and regeneration, puts PRF in a unique position as an autologous biological agent for the treatment of chronic inflammation, pain, and damages in joints and tendons, arising from injuries, arthrosis or arthritis.
Interacts on Inflammation
and the Sensory Pathways of Pain
_____________________

Pain in joints, tendons, ligaments and muscles, often arise from inflammation affecting the somatosensory nervous system1.
Upon injury, several cell types, including neurons and tissue-residing macrophages, produce soluble pro-inflammatory cytokines, and chemokines that activate surrounding cells3–7 and recruit circulating leukocytes, including macrophages, to the site of injury8–10.
Macrophages play a central role
Macrophages have an especially significant function in regulating inflammation and are considered to be common peripheral regulators of neuropathic pain5,10,11. Through the release of inflammatory mediators and interactions with neurotransmitters and receptors, the immune cells form an integrated network with the somatosensory system, coordinating the immune response, tissue healing and modulates the sensory pathways of pain8.
Showing Promising Results
in Cartilage Regeneration,
Meniscus and Tendon Repair
_____________________
Regeneration of Cartilage and Tendons
Cartilage and tendons are two of the most avascular and low cell density tissues found in humans, and thus have a very limited potential for repair and regeneration. Left untreated, defects often do not heal at all and stay in a chronic inflammatory state.
The capacity to modulate inflammation, influence macrophage polarization, and stimulate an acute response leading to accelerated healing and regeneration, puts PRF in a unique position as an autologous biological agent for the treatment of inflammation, pain, and damages in joints and tendons, arising from injuries, arthrosis or arthritis.

Additive-free liquid PRF
The introduction of the additive-free liquid PRF, which forms a fibrin clot upon injection, has been a proposed method for the regeneration of cartilage 18,19 and for accelerating the healing of tendons 38.
Full-thickness critical-sized osteochondral defects 5 mm in diameter and 5 mm in depth were created in the knee joint of 12 adult female New Zealand White rabbits. Defects were regenerated with either PRP or PRF and compared to control 18.
Biologically
Relevant Response
_____________________
Drop-by-Drop release – Combination of White Cells, Platelets
and Fibrin ensures a timely release of growth factors

Drop-by-Drop
In PRF, the delivery of cytokines and growth factors is done at a rate that is biologically relevant and responding to the needs of the environment7,20–25.
The function of fibrin has in this sense been compared to an irrigation system, where you do not want to drown the plants with large amounts of water in a single shot, but aim at delivering a biologically suitable amount of water and nutrients in a slow and adapted manner.
Cytokines
PDGF, VEGF, TGF-β EGF, fFGF, IGF-1, TGF-α, PAF, thrombospondin, platelet thromboplastin, coagulation factors, serotonin, histamine, hydrolytic enzymes and endostatin
Platelet-Derived Growth Factors
PDGFs (Platelet-Derived Growth Factors) are a family of essential and potent growth factors and chemotactic agents with an important role in wound healing39,40.
Recombinant PDGF is furthermore used in medicine to help heal chronic ulcers and in orthopedic surgery and periodontics as an alternative to bone autograft to stimulate bone regeneration and repair.
Vascular Endothelial Growth Factor
PDGF and VEGF (Vascular Endothelial Growth Factor) work together to establish and increase the blood supply of worn down and ischemic tissue, by promoting the formation of new blood vessels (angiogenesis)38 .
Transforming Growth Factor Beta
TGF-β (Transforming Growth Factor Beta) is a multi-functional factor important for, amongst other things, the regulation of the inflammatory response. It controls many things, from cell division, differentiation, and controlled cell death of many different cell types along with IGF-1 39,40, to the stimulation of fibroblast production of hyaluronic acid (HA)42 and collagen 41.
Bio-PRF Sportsmedicine
Next Generation PRF Solution
_____________________

Research-driven
Bio-PRF offers you the next level in blood-derived regenerative medicine. We are pushing the limits of Platelet Rich Fibrin based on in-depth research from our global research team. We are offering you solutions with significantly increased cell content and cell distribution as well as a wast extension of PRF resorption time all based on state-of-the-art research and technology.
Concentrated PRF (C-PRF) the new benchmark
As our research team gained insight on how cells distribute across a fibrin clot, a new protocol was formulated increasing the amount of Leucocytes and Platelets 10 fold from the standard injectable PRF protocols. The C-PRF protocol sets a new benchmark in what to expect from a Liquid PRF product and forms the foundation of exciting new products like bio-graft, bio-bone, and bio-filler.
Solid PRF membranes for wound healing
Our research team has created a novel method for evaluating and quantifying cell types in platelet-rich fibrin, which has led to key findings on cell content and distribution in a fibrin clot. This new understanding led to the development of the Bio-PRF fibrin clot protocol named Solid PRF. In this, the cell content of the fibrin clot is 4 times higher compared to standard fixed-angle centrifugation systems and protocols. Add to this a clotting sequence that creates a Solid PRF clot after 8 minutes of centrifugation.
Bio-Heat, extending the resorption properties
 One of the main limitations of PRF has been it’s short in vivo turnover rate. However, by combining knowledge about how the heating of platelet-poor plasma (PPP) denature and reorganize albumin, with the C-PRF protocol, our research team created the Bio-Heat technology setup that vastly widens the potential of PRF in regenerative medicine by extending resorption properties for up to 4–6 months.
One of the main limitations of PRF has been it’s short in vivo turnover rate. However, by combining knowledge about how the heating of platelet-poor plasma (PPP) denature and reorganize albumin, with the C-PRF protocol, our research team created the Bio-Heat technology setup that vastly widens the potential of PRF in regenerative medicine by extending resorption properties for up to 4–6 months.
Bio-PRF Protocols
and Future Perspectives
 C-PRF – a highly concentrated form of injectable PRF(10x), allowing even larger amounts of regenerative cells to be injected into joints and tendons.
C-PRF – a highly concentrated form of injectable PRF(10x), allowing even larger amounts of regenerative cells to be injected into joints and tendons.
 Solid PRF – autologous fibrin membranes encapsulating vast amounts of regenerative cells, slowly releasing pro-angiogenic cytokines to accelerate the healing of even problematic leg ulcers.
Solid PRF – autologous fibrin membranes encapsulating vast amounts of regenerative cells, slowly releasing pro-angiogenic cytokines to accelerate the healing of even problematic leg ulcers.
 BioHeat – The Bio-Heat system is the first commercially available heating system extending the resorption properties of PRF from 2-3 weeks to 4-6 months
BioHeat – The Bio-Heat system is the first commercially available heating system extending the resorption properties of PRF from 2-3 weeks to 4-6 months
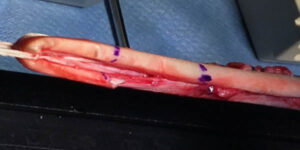
Solid PRF – used in replacement of anterior cruciate ligament where PRF membranes sutured into the harvested tendons during preparation to speed up healing time. combining the reservoir of different growth factors, cytokines, and lots of other molecules necessary for proper healing including various nutrients with suturable properties.

Liquid-PRF & Solid PRF – Combination enabling the production of PRF membranes of various sizes. These membranes are flexible and can be sutured offering new perspectives in wound healing and e.g. Gastrogastrointestinal surgery.

BioFiller – The ability to produce a slowly resorbable platelet-rich fibrin filler loaded with regenerative cells may offer an interesting new perspective in Sports Medicine and beyond, as a carrier of medicine or for filling cavities or prolonging the effect of joint injections.
Research Papers & Literature
The Evolution of PRF
To get an insight into the evolution of Platelet Rich Fibrin do visit “science behind” at www.bioprf.eu
Ongoing Research
To get an overview of the ongoing research in Bio-PRF please follow this link and see published and articles in progress.

Literature
- Wang, T. & He, C. Pro-inflammatory cytokines: The link between obesity and osteoarthritis. Cytokine Growth Factor Rev. 44, 38–50 (2018).
- Kiguchi, N., Kobayashi, D., Saika, F., Matsuzaki, S. & Kishioka, S. Pharmacological Regulation of Neuropathic Pain Driven by Inflammatory Macrophages. Int. J. Mol. Sci. 18, 2296 (2017).
- Kiguchi, N., Maeda, T., Kobayashi, Y., Fukazawa, Y. & Kishioka, S. Macrophage inflammatory protein-1α mediates the development of neuropathic pain following peripheral nerve injury through interleukin-1β up-regulation. Pain 149, 305–315 (2010).
- Mueller, M. et al. Rapid response of identified resident endoneurial macrophages to nerve injury. Am. J. Pathol. 159, 2187–2197 (2001).
- Thacker, M. A., Clark, A. K., Marchand, F. & McMahon, S. B. Pathophysiology of peripheral neuropathic pain: Immune cells and molecules. Anesthesia and Analgesia 105, 838–847 (2007).
- Zhang, F. F. et al. Perineural expression of high-mobility group box-1 contributes to long-lasting mechanical hypersensitivity via matrix metalloprotease-9 up-regulation in mice with painful peripheral neuropathy. J. Neurochem. 136, 837–850 (2016).
- Fujioka-Kobayashi, M. et al. Optimized Platelet-Rich Fibrin With the Low-Speed Concept: Growth Factor Release, Biocompatibility, and Cellular Response. J. Periodontol. 88, 112–121 (2017).
- Ren, K. & Dubner, R. Interactions between the immune and nervous systems in pain. Nature Medicine 16, 1267–1276 (2010).
- Scholz, J. & Woolf, C. J. The neuropathic pain triad: Neurons, immune cells and glia. Nature Neuroscience 10, 1361–1368 (2007).
- Kiguchi, N., Kobayashi, Y. & Kishioka, S. Chemokines and cytokines in neuroinflammation leading to neuropathic pain. Current Opinion in Pharmacology 12, 55–61 (2012).
- Ristoiu, V. Contribution of macrophages to peripheral neuropathic pain pathogenesis. Life Sciences 93, 870–881 (2013).
- Sánchez, M. et al. Plasma rich in growth factors to treat an articular cartilage avulsion: A case report. Med. Sci. Sports Exerc. 35, 1648–1652 (2003).
- Campbell, K. A. et al. Does Intra-articular Platelet-Rich Plasma Injection Provide Clinically Superior Outcomes Compared with Other Therapies in the Treatment of Knee Osteoarthritis? A Systematic Review of Overlapping Meta-analyses. Arthrosc. – J. Arthrosc. Relat. Surg. 31, 2213–2221 (2015).
- Kanchanatawan, W. et al. Short-term outcomes of platelet-rich plasma injection for treatment of osteoarthritis of the knee. Knee Surgery, Sport. Traumatol. Arthrosc. 24, 1665–1677 (2016).
- Meheux, C. J., McCulloch, P. C., Lintner, D. M., Varner, K. E. & Harris, J. D. Efficacy of Intra-articular Platelet-Rich Plasma Injections in Knee Osteoarthritis: A Systematic Review. Arthrosc. – J. Arthrosc. Relat. Surg. 32, 495–505 (2016).
- Nguyen, C., Lefèvre-Colau, M. M., Poiraudeau, S. & Rannou, F. Evidence and recommendations for use of intra-articular injections for knee osteoarthritis. Annals of Physical and Rehabilitation Medicine 59, 184–189 (2016).
- Sánchez, M. et al. Combination of Intra-Articular and Intraosseous Injections of Platelet Rich Plasma for Severe Knee Osteoarthritis: A Pilot Study. Biomed Res. Int. 2016, 4868613 (2016).
- Abd El Raouf, M. et al. Injectable-platelet rich fibrin using the low speed centrifugation concept improves cartilage regeneration when compared to platelet-rich plasma. Platelets 30, 213–221 (2017).
- Kazemi, D., Fakhrjou, A., Mirzazadeh Dizaji, V. & Khanzadeh Alishahi, M. Effect of Autologous Platelet Rich Fibrin on the Healing of Experimental Articular Cartilage Defects of the Knee in an Animal Model. Biomed Res. Int. 2014, 1–10 (2014).
- Dohan Ehrenfest, D. M., de Peppo, G. M., Doglioli, P. & Sammartino, G. Slow release of growth factors and thrombospondin-1 in Choukroun’s platelet-rich fibrin (PRF): a gold standard to achieve for all surgical platelet concentrates technologies. Growth Factors 27, 63–9 (2009).
- Choukroun, J. & Ghanaati, S. Reduction of relative centrifugation force within injectable platelet-rich-fibrin (PRF) concentrates advances patients’ own inflammatory cells, platelets and growth factors: the first introduction to the low speed centrifugation concept. Eur. J. Trauma Emerg. Surg. 1–9 (2017). doi:10.1007/s00068-017-0767-9
- Kobayashi, E. et al. Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin. Oral Investig. 20, 2353–2360 (2016).
- El Bagdadi, K. et al. Reduction of relative centrifugal forces increases growth factor release within solid platelet-rich-fibrin (PRF)-based matrices: a proof of concept of LSCC (low speed centrifugation concept). Eur. J. Trauma Emerg. Surg. 1–13 (2017). doi:10.1007/s00068-017-0785-7
- Dohan Ehrenfest, D. M. et al. The impact of the centrifuge characteristics and centrifugation protocols on the cells, growth factors, and fibrin architecture of a leukocyte- and platelet-rich fibrin (L-PRF) clot and membrane. Platelets 29, 171–184 (2018).
- Dohan Ehrenfest, D. M., Del Corso, M., Diss, A., Mouhyi, J. & Charrier, J. Three-dimensional architecture and cell composition of a Choukroun’s platelet-rich fibrin clot and membrane. J. Periodontol. 81, 546–55 (2010).
- Hom, D. B. New Developments in Wound Healing Relevant to Facial Plastic Surgery. Arch. Facial Plast. Surg. 10, 402–406 (2008).
- Sclafani, A. P. Applications of platelet-rich fibrin matrix in facial plastic surgery. Facial Plast. Surg. 25, 270–6 (2009).
- Martinez, F. O., Helming, L. & Gordon, S. Alternative Activation of Macrophages: An Immunologic Functional Perspective. Annu. Rev. Immunol. 27, 451–483 (2009).
- Parisi, L. et al. Macrophage Polarization in Chronic Inflammatory Diseases: Killers or Builders? J. Immunol. Res. 2018, 1–25 (2018).
- Nasirzade, J., Kargarpour, Z., Hasannia, S., Strauss, F. J. & Gruber, R. Platelet-rich fibrin elicits an anti-inflammatory response in macrophages in vitro. J. Periodontol. 91, 244–252 (2019).
- Zhang, J. et al. Anti‐inflammation effects of injectable platelet‐rich fibrin via macrophages and dendritic cells. J. Biomed. Mater. Res. Part A 108, 61–68 (2020).
- Arango Duque, G. & Descoteaux, A. Macrophage cytokines: involvement in immunity and infectious diseases. Front. Immunol. 5, 491 (2014).
- Opal, S. M. & DePalo, V. A. Anti-Inflammatory Cytokines. Chest 117, 1162–1172 (1999).
- Vogel, D. Y. S. S. et al. Human macrophage polarization in vitro: maturation and activation methods compared. Immunobiology 219, 695–703 (2014).
- Zhang, F. et al. TGF-β induces M2-like macrophage polarization via SNAILmediated suppression of a pro-inflammatory phenotype. Oncotarget 7, 52294–52306 (2016).
- Murray, P. J. et al. Macrophage Activation and Polarization: Nomenclature and Experimental Guidelines. Immunity 41, 14–20 (2014).
- Wong, C.-C. et al. Platelet-Rich Fibrin Facilitates Rabbit Meniscal Repair by Promoting Meniscocytes Proliferation, Migration, and Extracellular Matrix Synthesis. Int. J. Mol. Sci. 18, 1722 (2017).
- Sánchez, M. et al. Comparison of surgically repaired Achilles tendon tears using platelet-rich fibrin matrices. Am. J. Sports Med. 35, 245–251 (2007).
- Miron, R. J. et al. Platelet-Rich Fibrin and Soft Tissue Wound Healing: A Systematic Review. Tissue Eng. Part B. Rev. 23, 83–99 (2017).
- Ng, F. et al. PDGF, TGF-beta, and FGF signaling is important for differentiation and growth of mesenchymal stem cells (MSCs): transcriptional profiling can identify markers and signaling pathways important in differentiation of MSCs into adipogenic, chondrogenic, and o. Blood 112, 295–307 (2008).
- Sclafani, A. P. & McCormick, S. A. Induction of dermal collagenesis, angiogenesis, and adipogenesis in human skin by injection of platelet-rich fibrin matrix. Arch. Facial Plast. Surg. 14, 132–6 (2012).
- Kakudo, N. et al. Proliferation-promoting effect of platelet-rich plasma on human adipose-derived stem cells and human dermal fibroblasts. Plast. Reconstr. Surg. 122, 1352–60 (2008).





